|
A video demonstration of the illustration can be found below. 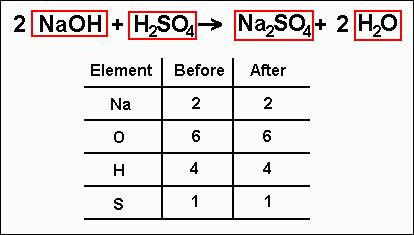
The illustration explores the process of balancing a chemical equation using the oxidation of a number of different hydrocarbons containing between one and five carbon atoms. The process of balancing an equation involves adding to each side of the equation until there are the same number of atoms of each element present on both sides. Without the proper number of reactants and products, a chemical equation is not a complete representation of the reaction. This is the significance of the numbers written before each compound in the reaction. A balanced chemical equation is one where there are the same number of atoms of each element on either side of the equation. Enter the chemical equation that is unbalanced because a chemical balancer will show an error message if the given equation is already in a balanced form. Or you can write your equation in the input box. How do we balance a chemical equation Count the number of atoms of the reactants and the products on both sides of the equation. To make sure that the equation conforms to the law of conservation of mass the equation must be balanced. To use the chemical equation calculator, simply make your equations by clicking on the elements in the above periodic table. A chemical reaction involves the rearrangement of atoms between molecules, not the creation or destruction of atoms. This mean that nothing can be gained or lost in the process. The law states that the mass of the reactants going into a reaction must be equal to the mass of the products. Properly written, the equation obey's the law of conservation of mass *. The chemical equation for the formation of sugar from water and carbon dioxide is: Numbers in front of each compound specify how many of each is required to convert all of the reactants to products. 
Since it is a portable software, you can carry it. In reactions with more than one reactant or product, plus signs separate the individual products and reactants from each other. Chemical Equation Balancer is a simple and portable balancing chemical equations calculator for Windows.An arrow separates reactants from products.Products - the substances formed by the reaction.Reactants - the substances transformed by the reaction.A properly formed chemical equation contains: This allows equations to provide detailed information about a reaction.Ī chemical equation can be thought of as a recipe for making a set of chemical compound using other compounds as starting material. Like mathematical equations, chemical equations conform to a set of rules. Chemists use equations to describe these interactions. Because adding more does not change the equilibrium, it is not accounted for in the expression.Of compounds interact with each other to form new compounds. For example, if more sugar is added to a solution after the equilibrium has been reached, the extra sugar will not dissolve (assuming the solution is not heated, which would increase the solubility). This is due to the fact that pure liquids and solids have no effect on the physical equilibrium no matter how much is added, the system can only dissolve as much as the solubility allows. This is because their activities are defined as one, so plugging them into the equation has no impact. 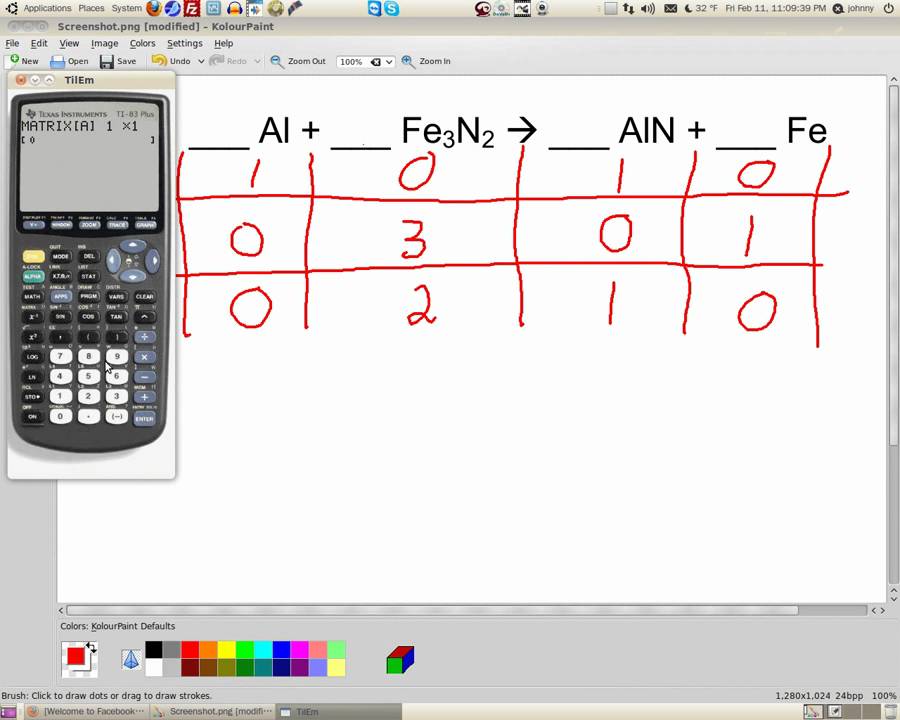
The lower case letters are the stoichiometric coefficients that balance the equation.Īn important aspect of this equation is that pure liquids and solids are not included. For K c, the activities are defined as the molar concentrations of the reactants and products (, etc.). The activities of the products are in the numerator, and those of the reactants are in the denominator.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
